Publication
Advanced Redox Technology Lab
Publication
Advanced Redox Technology Lab
Journal papers
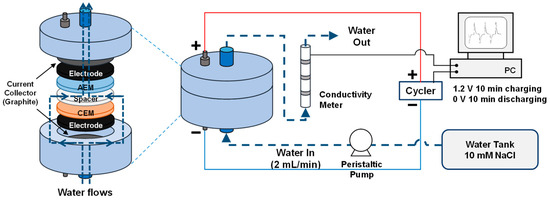
Membrane capacitive deionization (MCDI) is a modification of capacitive deionization (CDI) using ion-exchange membranes (IEM) in front of the electrodes. Electrode properties, especially the specific surface area, are known to be strongly related with desalination performance in CDI, but the effects of other properties in MCDI are not fully understood. The objective of this study was to investigate the effect of hydrophilicity in activated carbon electrodes on desalination performance in MCDI. Two types of activated carbon (P60 and YS-2) whose specific surface areas were similar were used as electrode materials, but they had different hydrophilicity (i.e., P60 was originally hydrophobic and YS-2 was relatively hydrophilic due to its nitrogen-containing surface chemistry). These hydrophilic electrodes (either the electrode itself or modified with polydopamine (PDA)) led to an increase in the salt adsorption capacity (SAC) in MCDI because they facilitated the access of both ions and water molecules into the electrode pores. In particular, the SAC of the P60 electrode displayed a large increase to almost reach that of the YS-2 electrode due to the improved hydrophilicity with PDA modification and the insignificant effects of PDA modifi취소cation on an already hydrophilic YS-2 electrode. Additionally, PDA-modified IEM in MCDI reduced the SAC as a result of the additional insulating PDA layer with little changes in hydrophilicity.