Publication
Advanced Redox Technology Lab
Publication
Advanced Redox Technology Lab
Conference Abstract
As global importance of fresh water continues to rise, water hardness has emerged as a significant concern. Hard water, primarily originating from the chemical composition of ground water, is characterized by the presence of Ca2+ and Mg2+ ions, which cause hardness [1]. These ions can cause scaling in pipes [1] and diminish the cleansing efficiency of soaps [2]. Currently, the removal of these hardness ions is predominantly achieved through ion exchange methods, which necessitate chemical regeneration processes and may produce environmentally harmful by-products [1]. Therefore, to mitigate operational inefficiencies and environmental impacts, chemical-free electrochemical methods are being proposed as alternatives.
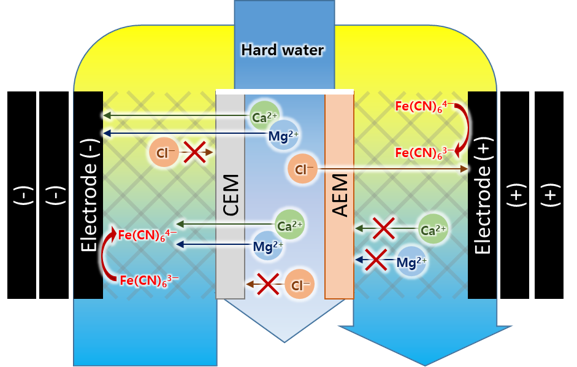
Among electrochemical ion removal methods, capacitive deionization (CDI) and electrodialysis (ED) are prominent, and a further advancement is the redox flow system. The redox flow system offers significant advantages, as it can operate at low voltages while utilizing reversible redox couples for energy recovery [3]. Although numerous previous studies have concentrated on the desalination of monovalent ions [3, 4], few researchers have focused on the removal of divalent ions such as Ca2+ and Mg2+ using the redox flow system. In this study, the redox flow system is utilized in a water softening system, and its performance under real hard water conditions was evaluated. Additionally, the transport mechanism of hardness ions was analyzed across various operational conditions of the system. The results demonstrate that the redox flow system effectively reduces the concentration of Ca²⁺ and Mg²⁺ ions in hard water.
The redox flow system allows for continuous removal of Ca2+ and Mg2+ ions due to the creation of an electric field resulting from the electrical imbalance generated by redox couple reactions. This system can continuously produce fresh water over extended periods without requiring a discharging step, unlike the CDI system. This continuous operation has also been reported in previous studies utilizing the system for Na+ ion removal. [5] In previous study, the transfer of Na+ ions through the ion-exchange membrane from the side channel to the mid-channel was considered negligible. [4] However, this was not the case in our study. The Na+ concentration in the mid-channel feed of our system was so low that the amount diffused from the side channel was significantly higher. Therefore, we conducted not only conductivity but also individual ion concentration analysis using ion chromatography, incorporating into charge efficiency calculations.
Our experiments varying the flow rate indicated that a higher flow rate results in higher charge efficiency, although the removal of hardness ions is reduced. This suggests that the removal of Ca2+ and Mg2+ ions requires a relatively longer residence time compared to Na+ ions. Voltage experiments demonstrated that the highest charge efficiency is achieved at an applied voltage of 1.2 V. Additionally, total hardness removal did not increase when the voltage was raised above 1.2 V. This plateau effect indicates that the system reaches its optimal ion removal capacity at this voltage, and further increasing the voltage may lead to undesirable side reactions or increased energy consumption without additional benefits in ion removal. By analyzing the individual ion concentrations, we were able to predict the behavior of each ion and gain insights for identifying the optimal conditions for this system. Furthermore, the redox couple provided a stable and reversible reaction environment, facilitating continuous operation without significant degradation. This stability is essential for maintaining long-term operational efficiency and minimizing maintenance requirements.
Overall, the insights gained from our experiments provide a comprehensive understanding of the factors influencing ion removal in the redox flow deionization system. These findings can guide the optimization of operational parameters, such as flow rate and applied voltage, to achieve the most efficient and effective water softening performance. Additionally, the analysis of individual ion behaviors can help in designing tailored solutions for specific water hardness issues, ensuring reliable and sustainable water treatment processes.